This is an archive copy of the IUCr web site dating from 2008. For current content please visit
https://www.iucr.org.
![[IUCr Home Page]](/iucr-top/logos/iucrhome.gif)
![[Commission Home Page]](/iucr-top/logos/cteach.gif)


Next: The Negative Quartet Relation
Up: An Introduction to Direct Methods. The
Previous: The Triplet Relation from Sayre's Equation
The triplet relation, although a two-dimensional phase relation, is very
successful in solving three-dimensional crystal structures. Nevertheless, it
may be more appropriate to try to solve structures with three-dimensional phase
relationships, the quartet relations.
The positive quartet relation is formulated as:
| 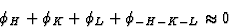 |
(32) |
for large E4 = N-1|EHEKELE-H-K-L|.
Analogous to the treatment of the triplet relation now three strong reflections
H, K and L are combined and the electron density must be found in the sets
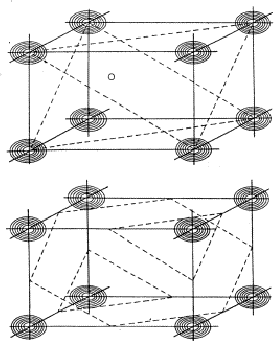
of planes of Fig. 12. As a result the electron density will be found near the
points of intersection of the three planes which are indicated for only a few
planes from the sets in Fig. 13. For a strong reflection -H -K -L it is
much more likely that its plane of maximum electron density will run through the
points of intersection (Fig. 14a) than that it will clear these points (Fig.
14b). From Fig. 14a the quartet relation (32) follows as straightforward as the
triplet relation from Fig. 10a (Schenk, 1981).
Figure 12:
If the reflections H, K and L are strong, the electron
density will probably lie in the neighbourhood of the three sets of
equidistant planes defined by H, K and L.
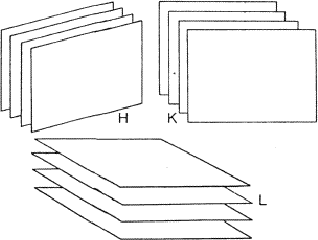 |
Figure 13:
The electron density will be found near the points of intersection
of the three sets of planes H, K and L.
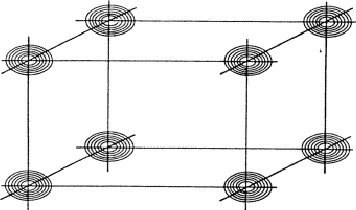 |
Figure 14:
If reflection -H-K-L has also a large intensity, it is more
likely that its planes of maximum intensity run through the points of
intersection of H, K and L (a) than that they run clear of them (b).
 |
This quartet relation, however, is not as strong as the triplet relation because
of the factor N-1 in E4. It will be recalled that in E3 a term
N-1/2 appears. The reliability is improved by combining the quartet
with an identical one constructed from two triplets:
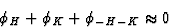
| 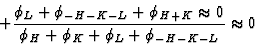 |
(33) |
which holds for large E4 and large |EH+K|. This can be understood by
drawing the H+K reflection in Fig. 13, as indicated in Fig. 15a. Reflection
H+K strong indicates that the electron density will be found near the
intersection of H and K and thus a large |EH+K| is an additional
indication that quartet (32) is true. In Figs. 15b and 15c similar situations
are sketched for the other two cross terms H + L and K + L, which both
leads to sums of triplets analogous to (33) involving the phases 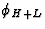 and
and 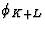 respectively. So in conclusion a large value of E4 and
large |EH+K|, |EH+L| and |EK+L| are indications that the positive
quartet relation (32) is likely to be true, and thus positive quartets are
controlled by the magnitudes of 7 structure factors.
respectively. So in conclusion a large value of E4 and
large |EH+K|, |EH+L| and |EK+L| are indications that the positive
quartet relation (32) is likely to be true, and thus positive quartets are
controlled by the magnitudes of 7 structure factors.
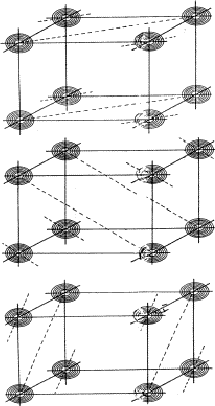
Figure 15:
If H, K, L and -H-K-L are strong and the electron density
will be found near their points of intersecting, the H + K reflection,
indicated in (a) with a dotted line, is expected to be strong, conversely a
large |EH+K| supports the quartet relation (11). Figs (b) and (c)
give the analogous situations with respect to the H + L and K + L
reflections.
 |


Next: The Negative Quartet Relation
Up: An Introduction to Direct Methods. The
Previous: The Triplet Relation from Sayre's Equation
Copyright © 1984, 1998 International Union of
Crystallography
IUCr
Webmaster
![[IUCr Home Page]](/iucr-top/logos/iucrhome.gif)
![[Commission Home Page]](/iucr-top/logos/cteach.gif)
![]()
![]()
![]()



![]()
